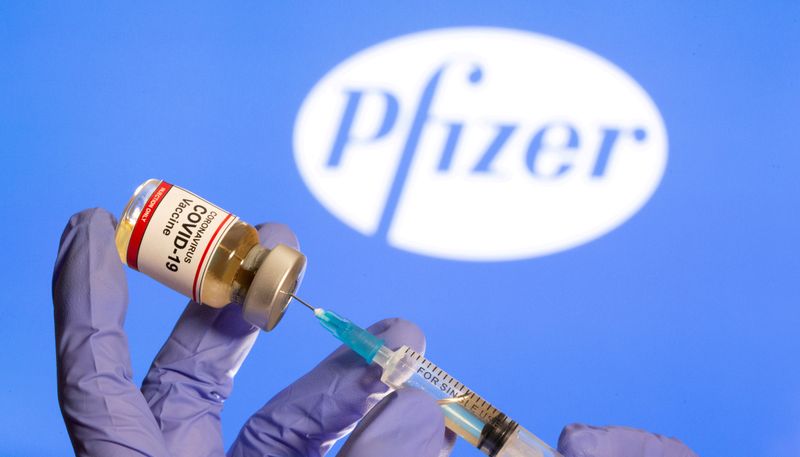
(Reuters) – An Alaskan health worker had a serious allergic reaction after getting Pfizer Inc and BioNTech’s coronavirus vaccine, but is now stable, public health authorities said on Wednesday.
The adverse reaction in the person, minutes after taking the Pfizer shot on Tuesday, was similar to two cases reported last week in Britain.
Britain’s medical regulator has said that anyone with a history of anaphylaxis, or severe allergic reactions to a medicine or food, should not get the Pfizer-BioNTech COVID-19 vaccine.
But the U.S. Food and Drug Administration has said that most Americans with allergies should be safe to receive the vaccine. It said only people who have previously had severe allergic reactions to vaccines or ingredients in this particular vaccine should avoid getting the shot.
The Alaskan patient did not have a history of allergic reactions, Lindy Jones, the director of the emergency department in the capital Juneau where the patient was treated, told reporters at a virtual briefing.
The symptoms in the middle-aged patient resolved after being administered with allergy treatment epinephrine, Jones said.
The patient was still in Juneau’s Bartlett Regional Hospital being monitored on Wednesday.
Pfizer said the vaccine comes with a clear warning that appropriate medical treatment and supervision should always be readily available in case of anaphylaxis, but it would update the labeling language for the vaccine if needed.
Administration of the vaccine began Monday in the United States, following emergency-use authorization last week. Early doses have been set aside for healthcare workers and nursing home residents
Former FDA Chief Scientist Jesse Goodman called the allergic reaction concerning but said that more information must be known in order to better understand the risks.
“What we need to know is what the denominator is — how many doses have been given? Is this going to be something that’s going to be seen at a higher incidence with this vaccine than with others?” Goodman said. “We’re going to have to find out those things to inform whether that changes recommendations or how this is used.”
(Reporting by Manas Mishra in Bengaluru; Additional reporting by Michael Erman in New York; Editing by Ramakrishnan M. and Shinjini Ganguli and Alistair Bell)