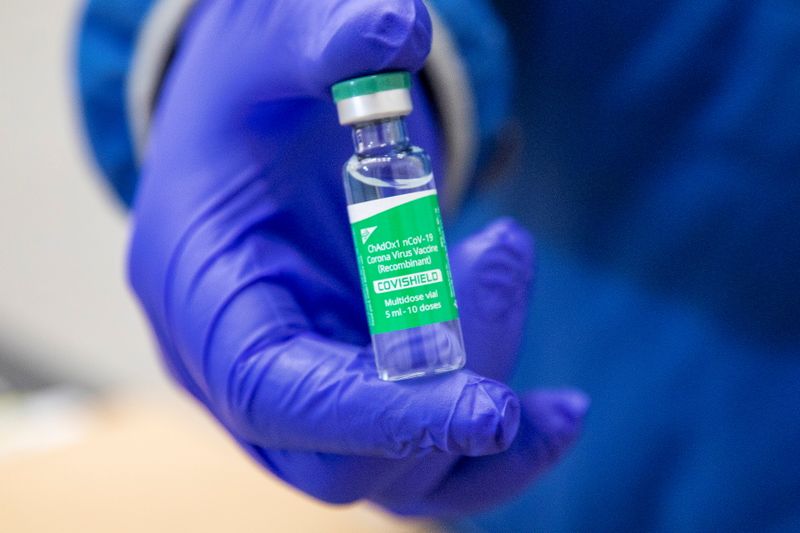
OTTAWA (Reuters) – Canada’s health ministry said on Wednesday it would not restrict use of AstraZeneca Plc’s COVID-19 vaccine after a review showed the benefits outweighed the very rare risk of blood clots.
A separate advisory council had earlier recommended Canada stop offering the vaccine to people under 55. The panel is now reviewing that advice, the health ministry said in a statement.
Denmark on Wednesday became the first country to stop using the vaccine altogether over a potential link to the rare blood clots. Other nations have imposed limits on its use.
But Health Canada, the federal health ministry, said in a statement that a review of data from Europe, Britain and AstraZeneca had not identified specific risk factors.
“Therefore, Health Canada is not restricting the use of the vaccine in any specific populations at this time … The potential risk of these events is very rare, and the benefits of the vaccine in protecting against COVID-19 outweigh its potential risks,” it said.
Canada on Tuesday said it had recorded its first case of blood clotting with low platelets after someone received the AstraZeneca shot. The patient in question, a woman from Quebec, is recovering. (Graphic on vaccines: https://tmsnrt.rs/3tUM8ta)
COVID-19 cases are surging in Canada with the country reporting a near-record number of new cases recently. (Graphic on cases: https://tmsnrt.rs/34pvUyi)
(Reporting by David Ljunggren in Ottawa and Allison Martell in Toronto; Editing by Lisa Shumaker)