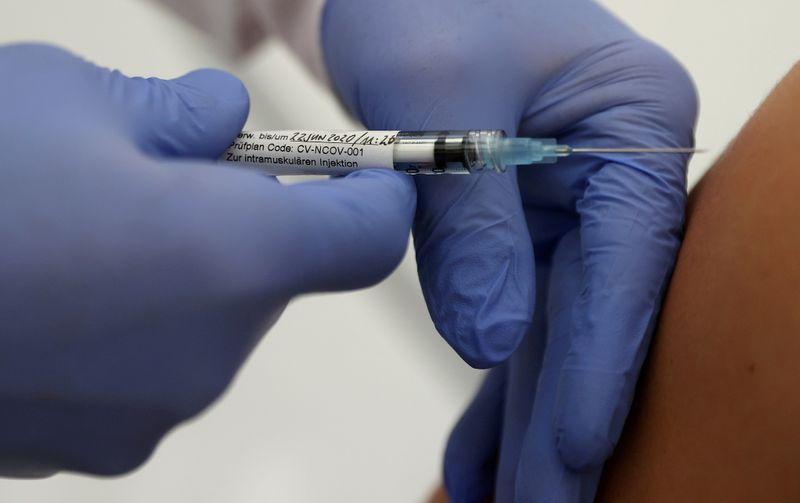
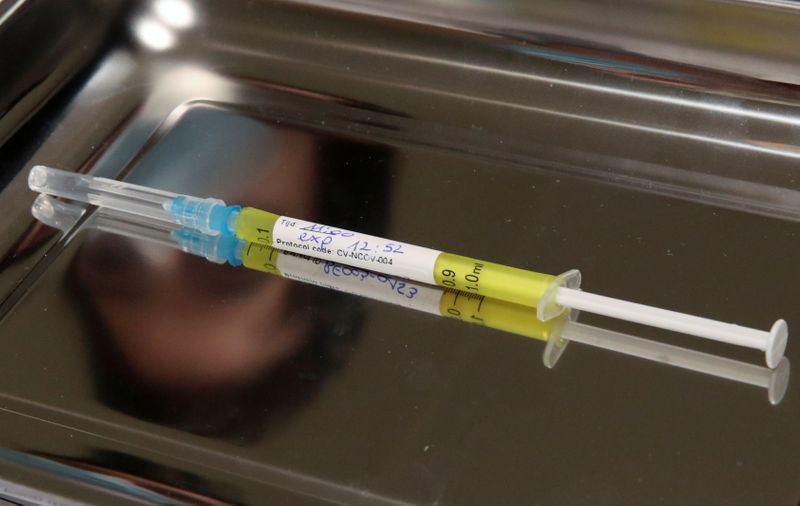
BERLIN (Reuters) – Germany’s CureVac, which is gearing up to publish results of a key COVID-19 vaccine trial, said U.S. export restrictions on key materials are making it impossible to predict its short-term supply ramp-up in Europe.
“Due to the Defense Production Act we are not getting certain goods out of the USA,” CureVac Chief Executive Franz-Werner Haas told weekly Der Spiegel.
The Defense Production Act is a decades old U.S. law that gives federal agencies the power to prioritize procurement orders related to national defence, but it has also widely been used in non-military crises such as natural disasters.
“We are not getting all the materials that we need,” he was quoted as saying.
“At times we live from hand to mouth. That makes it hard to build up a stockpile,” Haas said, when asked how much the company would be able to deliver during the summer months.
He added that items in short supply include special plastic containers and nucleotides, which are the chemical building blocks that make up the genetic code in the vaccine’s active ingredient, the so-called messenger RNA.
Unlike rival German vaccine developer BioNTech, which struck a partnership with pharma giant Pfizer, CureVac has not teamed up with a U.S. partner. BioNTech and Pfizer continue to increase global production and have not cited the Defense Production Act as a hindrance.
As CureVac’s only major supply deals, the European Union in November last year secured up to 405 million doses of the immunisation.
The biotech firm has said it expects to file for European authorization in late May or early June.
Nasdaq-listed CureVac, which is backed by investors Dietmar Hopp, GlaxoSmithKline as well as the German government, has said it aims to produce up to 300 million doses of the vaccine in 2021 and up to 1 billion in 2022.
(Reporting by Ludwig Burger)