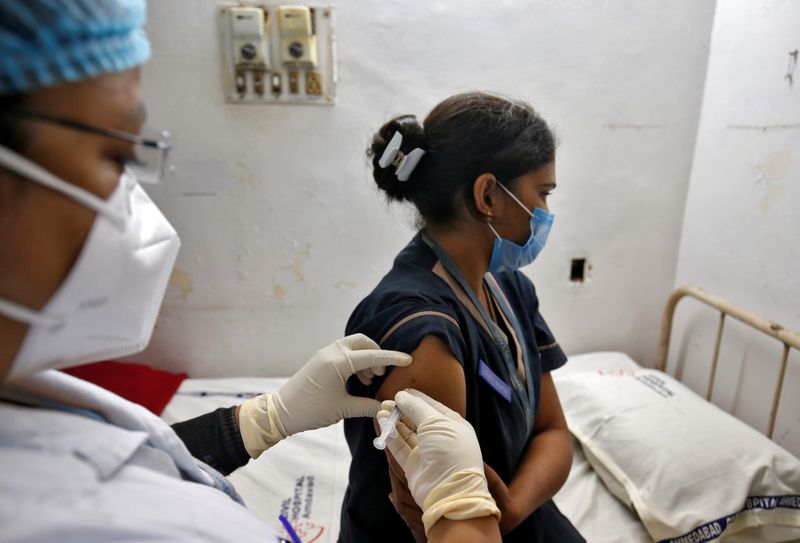
NEW DELHI/BENGALURU (Reuters) – Serum Institute of India (SII) and Bharat Biotech said on Tuesday they would end a public spat and focus on rolling out COVID-19 shots after executives of both Indian vaccine makers clashed over the approval of their rival shots.
“Both companies respect the great work being carried out by each other and put behind us the miscommunication and misunderstanding caused during the past week,” SII’s Chief Executive Officer Adar Poonawalla and Bharat Biotech’s Chairman Krishna Ella said in a joint statement.
India’s drugs regulator on Sunday approved for emergency use a vaccine developed by AstraZeneca and Oxford University, which will be manufactured by SII, and another developed domestically by Bharat Biotech.
But the approval for Bharat Biotech’s COVAXIN, developed with a government research body, faced criticism from industry experts and others as the company has yet to release efficacy and safety data, and it still has a Phase III trial underway.
“Until we get the efficacy result, how do we know that anything works?” Poonawalla told Reuters on Sunday.
Ella, however, defended both his firm and the approval for the vaccine in an online press conference on Monday.
“I want to say that it hurts as scientists. We work 24 hours. Do we deserve that type of bashing from the people?” Ella said, adding his firm was being picked on because it was Indian.
Ella also took a swipe at AstraZeneca, which said in November a dosing error had led to promising results in a subset of its trial volunteers.
In Tuesday’s joint statement, the companies said they were fully aware of the importance of vaccines for people and countries and they jointly pledged to provide global access to their COVID-19 vaccines.
(Reporting by Devjyot Ghoshal and Sachin Ravikumar; Additional reporting by Nivedita Bhattacharjee and Anuron Mitra; Editing by Euan Rocha and Mark Potter)