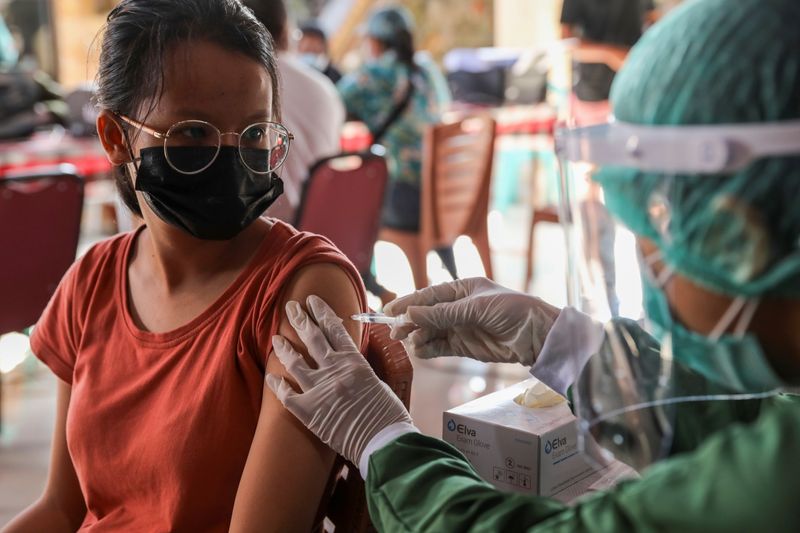
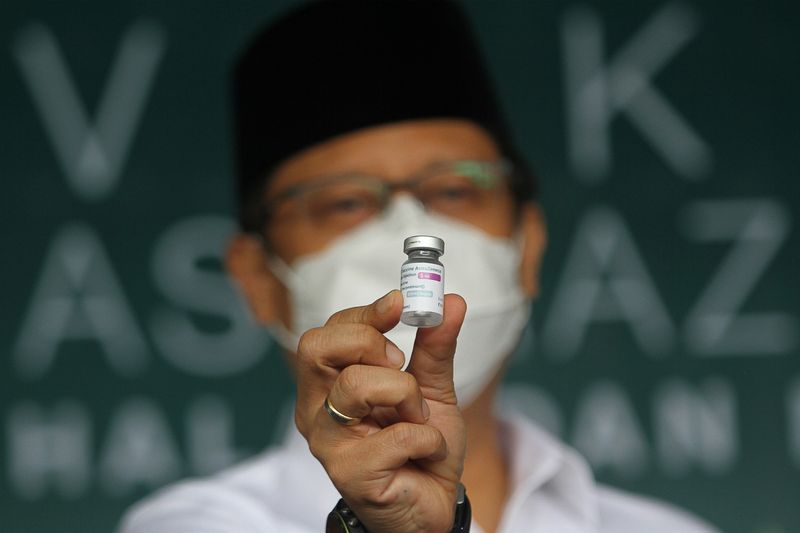
SURABAYA, Indonesia (Reuters) – The rollout of AstraZeneca’s COVID-19 vaccine has started in Indonesia, the world’s largest Muslim majority nation, despite concern and some confusion over whether the Anglo-Swedish product is halal, or permissible under Islam.
The country has started distributing the vaccine in six provinces, a week after the Indonesia Ulema Council, the main Muslim group that issues halal certifications, last week said it contained trypsin sourced from the pancreas of a pig.
AstraZeneca has said the vaccine contains no pork-derived ingredients.
The East Java chapter of the Ulema council on Monday said the vaccine was halal when it was administered to some of its members. It did not say why it had been deemed halal.
Misbakhul Munir, a 57-year-old member of the Nahdlatul Ulama in East Java, said he welcomed the AstraZeneca vaccine, but laments the mixed messages about it.
“I strongly dislike the ambiguous decision,” he said of the halal status. “I’ll just adhere to what the local council said.”
Indonesia gave the go-ahead to use the AstraZeneca vaccine on Friday after European experts found no link between the vaccine and reports of blood clots among some its recipients in Europe.
Siti Nadia Tarmizi, a senior Indonesian health ministry official, told Reuters about 100 religious figures in East Java had received coronavirus vaccines on Tuesday, either those of AstraZeneca or Sinovac Biotech.
She added there’s been no vocal opposition to the AstraZeneca vaccine.
Indonesia’s immunisation programme started in January and aims to reach 181.5 million people within a year. The country is suffering one of the worst COVID-19 epidemics in Asia, with 1,471,225 cases and 39,865 deaths as of Tuesday.
Indonesia has secured over 426 million doses of vaccines in total, including from Pfizer and BioNTech, Novavax, AstraZeneca and Sinovac.
(Writing and additional reporting by Stanley Widianto in Jakarta; Editing by Martin Petty and Chizu Nomiyama)