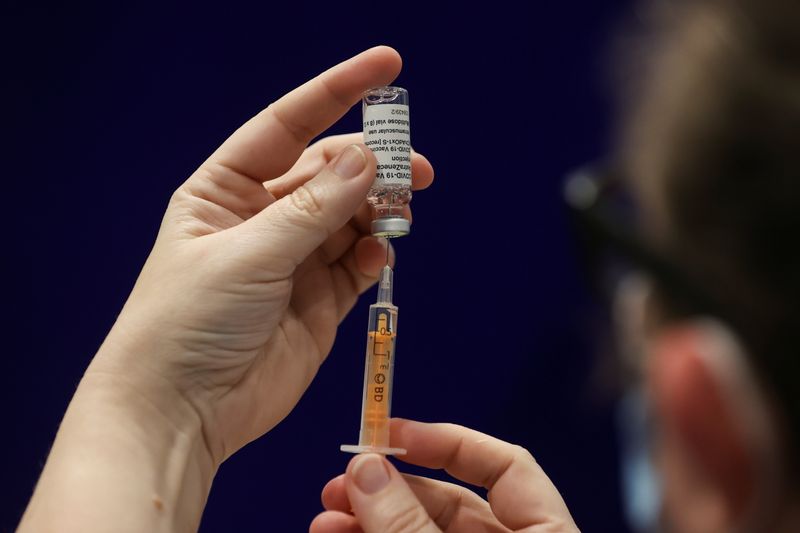
ROME (Reuters) – Italy’s medicines regulator AIFA said on Saturday it had approved the use of the COVID-19 vaccine produced by AstraZeneca and Oxford University, a day after the green light by the European Medicines Agency (EMA).
It recommended preferential use of the vaccine for adults aged 55 and under.
The vaccine is the third cleared for use in the European Union after the ones produced by Pfizer and BioNTech, and by Moderna.
“The arrival of a third vaccine is an important contribution to the ongoing vaccination campaign,” AIFA Director General Nicola Magrini said.
AIFA’s technical-scientific commission confirmed the AstraZeneca vaccine had an efficacy of around 60% and was “a valid additional option” in combating the pandemic, the statement said.
The regulator added that given study data showed a level of uncertainty in assessing how the vaccine worked for people over the age of 55 it recommended “the preferential use” of the vaccine in people between the ages of 18 and 55.
On Friday the EMA had said there were not yet enough results for people over the age of 55 to determine how well it worked in that group. But it said the vaccine could still be given to older people.
(Reporting by Giulia Segreti; Editing by Frances Kerry)