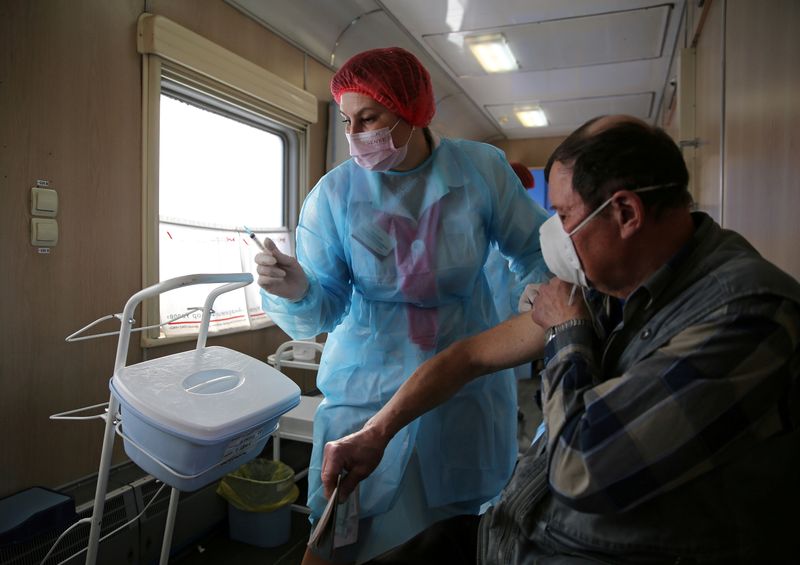
MOSCOW (Reuters) – Some of those who volunteered for the clinical trial of Russia’s second COVID-19 vaccine have broken rank and taken part in go-it-alone “citizen experiments” which they say raise concerns about the shot’s efficacy.
A group including 120 former participants in the trial for the EpiVacCorona vaccine, developed by the Vector Institute in Siberia, ran commercially available antibody tests and sent frozen blood plasma samples to independent laboratories with the aim of testing the vaccine’s ability to neutralise an infection.
“We have serious concerns about the efficacy of the peptide vaccine EpiVacCorona,” the group, which is led by businessman Andrey Krynicki, said in an open letter to the health ministry published on their blog on Wednesday.
“The results of our tests diverge from what… officials have said in the media, which is why we are concerned.”
The scientists behind the vaccine, which has been approved by the Russian regulator and added to the national inoculation programme, say it is safe and effective, and question the scientific viability of the group’s findings.
EpiVacCorona, Russia’s second vaccine to be registered, is a synthetic peptide vaccine which uses a different technology from the better-known Sputnik V shot.
Unlike for Sputnik V, whose late-stage, Phase III trial results were published in The Lancet medical journal in February, developers of Russia’s second shot have not yet disclosed larger scale trial results.
Yet more than 115,000 doses have been deployed in the national inoculation programme, according to authorities. Over 5 million doses should be ready by July.
Asked to comment on the letter, the head of the Yekaterinburg branch of the Vector Institute, Alexander Semenov, said scientists were racing to publish Phase III results for EpiVacCorona and they would be ready very soon.
He pointed to results from early-stage, Phase I/II trials, published in an academic journal, in which the vaccine developers said the shot was tested on 100 people and found to be 100% effective in producing antibodies “specific to the antigens that make up the vaccine”.
ANTIBODY TESTS
The open letter’s authors said that the group, whose 19 other members are people who received the vaccine as part of the nationwide inoculation drive, took antibody tests of which 30% were negative. They said the tests used had been recommended to by the vaccine’s developers.
By contrast, Semonov said that average and commonly available testing kits are not specific to EpiVacCorona, and so may not reveal the antibodies it produces. This applies to all peptide-based vaccines globally, he said.
Semonov also said that without proper data on the methods used and or proper scientific analysis, it was impossible to say if the group’s findings were correct.
“The signatories of the open letter to the health ministry must present the protocols of the laboratory experiments that they conducted,” he added. “So far, I have not seen any.”
He said that the vaccine provides protection from COVID-19 only once both shots have been administered, starting from around 45 days after the first dose.
Out of Russia’s population of 145 million, 4.3 million people have now been fully vaccinated against COVID-19, almost all with Sputnik V, including around 1 million in the capital, according to officials.
(Reporting by Polina Ivanova; Editing by Kate Kelland and Raissa Kasolowsky)